Attached is the latest publication on the Saudi Food and Drug Authority (SFDA) and the work they do as the regulator of food, drugs and medical devices in Saudi Arabia, they play a vital role in protecting and promoting public health, both in the Kingdom and internationally. In this publication, you will learn about the SFDA’s role as a regulatory authority, as well as their efforts to increase food and drug safety in support of Saudi Arabia’s goal of creating healthier citizens.
The publication covers the following key issues:
SFDA in Brief and their achievements for in 2020 giving more insights how they will be moving forward this year and the coming years, where the number of days needed to register products decreased dramatically between 2018 and 2020 as shown in the following Figure:
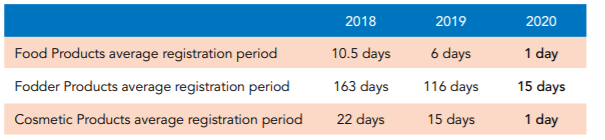
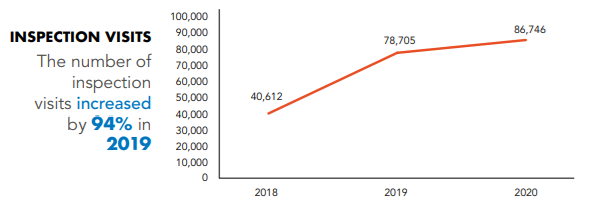
In addition, to the achievement on the Inspections and Product Registrations levels, having the number of inspection visits increased by 94% in 2019 as shown in the below enclosed Figure.
The most interesting section is the SFDA vision for 2030 and what it holds in the future for the pharmaceutical and food industry where it targets the industrial expansion and investment support. This includes localising key industries and increasing the local manufacture of essential products. The National Industrial Strategy (NIS) has identified pharmaceuticals, medical supplies and food processing as key segments for domestic manufacturing expansion. By 2030 the NIS is targeting the development of a pharmaceutical, biopharmaceutical and medical supplies industry, and the development of industrial clusters for food. This might be a future opportunity or a threat to international pharmaceutical industries. whether to localize their manufacturing within the SFDA or keep the full importation model.
Attached is the full publication for your review, please don't hesitate to reach out if further clarification is needed.
Asma Hammad